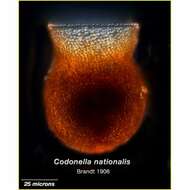
-
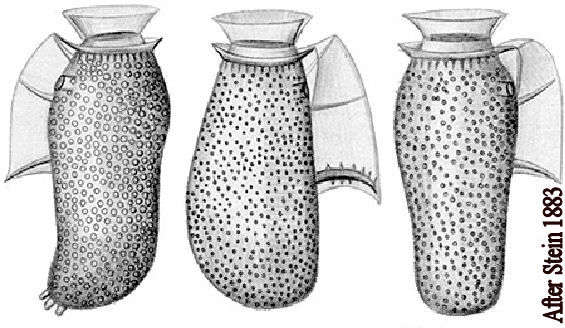
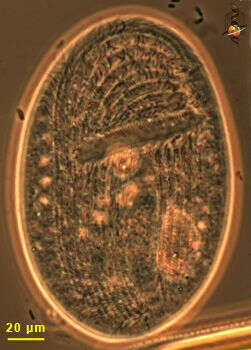
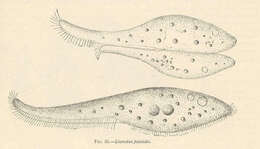
Fig 1: Dinophysis sacculus Schematic diagram (ventral view)from Stein 1883.
-
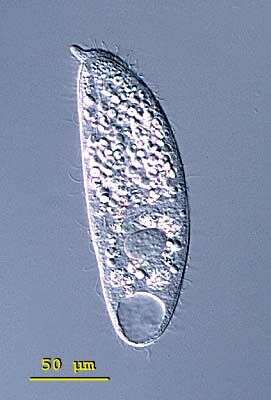
Spathidium (spa-thid-ee-um) porculus, the body is elongate, the posterior end is bluntly pointed or rounded but the anterior end is distinctively swollen - often fan-shaped and obliquely truncated. There is an ciliated apical ridge which is lined by toxicysts. The oral aperture is a slit that lies along the length of this ridge. The cilia are uniformly distributed in longitudinal parallel rows on both lateral surfaces. The macronucleus is highly variable, often elongate, ribbon-like or moniliform. The contractile vacuole is single and at the end of the cell. Spathidium feeds on other ciliates. It lives in fresh water ponds and lakes. The species is unmistakable because of its nose like projection of the oral apparatus. Slightly squashed cell. 136 microns. Differential interference contrast.
-
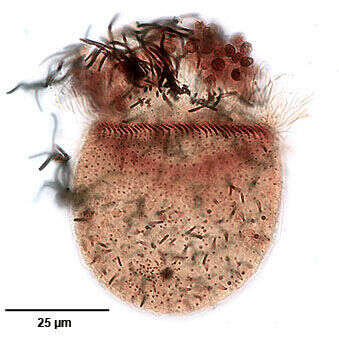
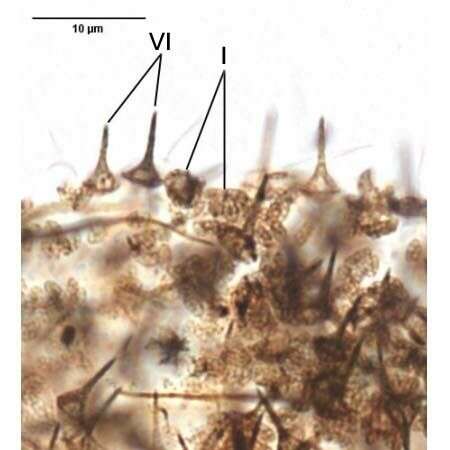
Ventral infraciliature of the haptorid ciliate, Monodinium balbianii (Fabre-Domergue, 1888). The dorsal side is distinguished by the five parallel rows of clavate (club-shaped) cilia (the dorsal brush) just posterior to the anterior wreath of ciliated basal bodies. The longitudinal files of somatic kinetosomes (seen here) are unciliated except for the dorsal brush (not seen in this ventral view) and the obliquely inclined closely spaced ciliated basal bodies that form the ciliary girdle. Darkly stained extrusomes are visible here. Collected from a freshwater pond near Boise, Idaho March 2005. Stained by the silver carbonate technique (see Foissner, W. Europ. J. Protistol., 27:313-330;1991). Brightfield.
-
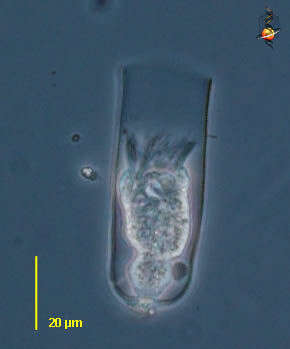
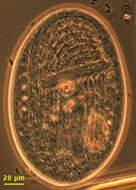
Undella (un-dell-a) is a tintinnid ciliate, in which the ciliate lives in a lorica. Species of Undella have a simple lorica, no adhering material, pedicels etc. The cell has contracted into the lorica, but the cilia of the adoral zone of membranelles are evident. Phase contrast.
-

Palustrimonas yorkeensis (Ruinen, 1938) Patterson and Simpson, 1996. Cell 8 -14 microns long, body not plastic and ovate to slender elliptical in profile. Anterior end may be somewhat pointed. The flagella insert sub-apically and are opposed. The anterior flagellum inserts into a short anteriorly directed pocket. The posterior flagellum inserts into a groove that extends for most of the length of the cell. The groove is often an elongated tear-shape with the point posterior-most. The anterior flagellum is approximately equal to the cell body in length and flails around the anterior end of the cell. The posterior flagellum is 2-2.5 times cell length and may be held fairly stiffly or beats in slow waves. The posterior flagellum generally appears thicker than the anterior flagellum. In some cells the posterior flagellum is missing. Swimming is often erratic and the cells tumble such that progress in one direction is quite slow. The cells do not glide. Many cells had large colourless inclusions in the posterior end of the cell suggesting that P. yorkeensis is predatory. In some cells the inclusions distended the cell sufficiently to obliterate the posterior portion of the groove. One cell was observed with an inclusion in the apical region which suggests that food may be ingested at the anterior end of the cell.
-
-
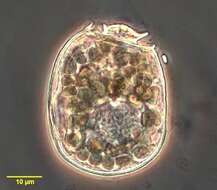
This is the ventral face of the ciliate showing kineties running around the mouth. A few somatic kineties run uninterrupted to the right of the cytostome (left in this picture as we are looking at the ventral face) arching around the anterior of the cell. Several right somatic kineties are interrupted by the cytostome. A long membrane of cilia lies anterior to the mouth. Gastronauta feeds mainly on diatoms. Phase contrast illumination
-
Two of the three types of lepidosomes of Luporinophrys micelae (FOISSNER,2005). The type II are not seen in this image.Stained by the silver carbonate technique (see Foissner, W. Europ. J. Protistol., 27:313-330;1991).Brightfield.
-
This tintinnid was found in a sample from the Eastern Mediterrean Sea in September
-
Sinophysis microcephalus observed in marine muds and sandy sediments in the vicinity of Broome, Western Australia in September 2003. This image was taken using phase contrast optics. This work was supported by the Australian Biological Resources Study.
-
Ventral view of Aspidisca lynceus (MUELLER,1773) EHRENBERG, 1830.Collected from a freshwater irrigation canal in Boise,Idaho December 2007.DIC
-
Almind Sø, Jylland, Danmark
-
Centers for Disease Control/Division of Parasitic Diseases and Malaria
EOL staff
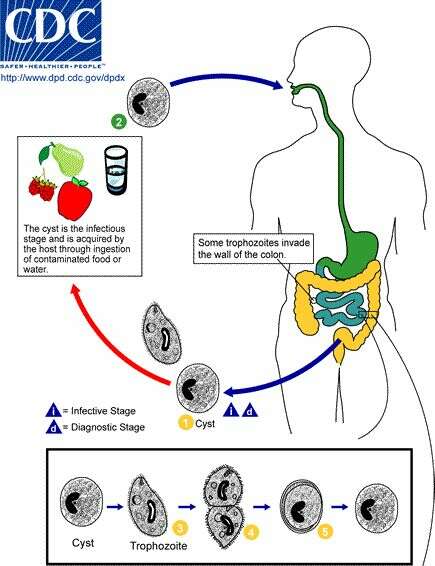
Life cycle of Balantidium coli, the cause of human balantidiasis The cyst stage (1) of the B. coli life cycle is responsible for transmission.The host most often acquires the cyst through ingestion of contaminated food or water (2). Following ingestion, excystation occurs in the small intestine and the trophozoites colonize the large intestine (3). The trophozoites reside in the lumen of the large intestine of humans and other animals, where they reproduce by binary fission, during which conjugation may occur (4). Trophozoites undergo encystation to produce infective cysts (5). Some trophozoites invade the wall of the colon and multiply. Some return to the lumen and disintegrate. Mature cysts are passed with feces (1).From
Centers for Disease Control Parasites and Health website
-
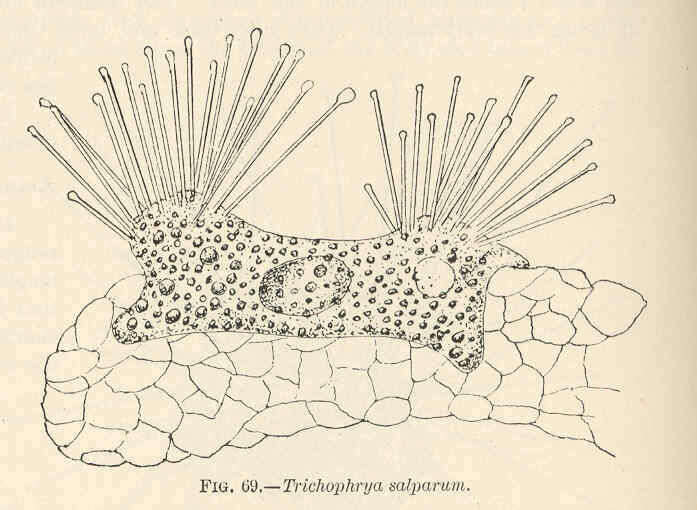
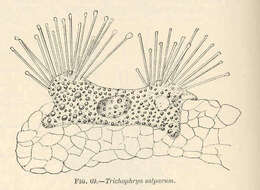
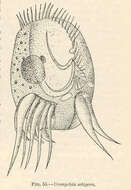
Trichophrya salparum.
-
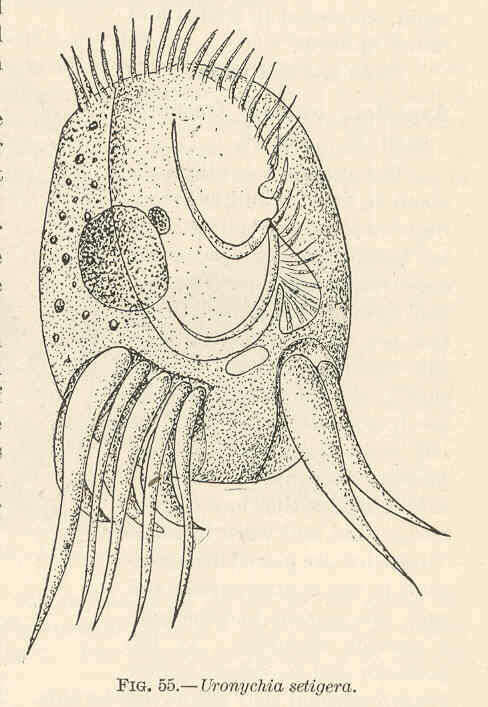
Uronychia setigera.
-
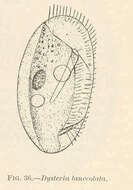
Dysteria lanccolata.
-
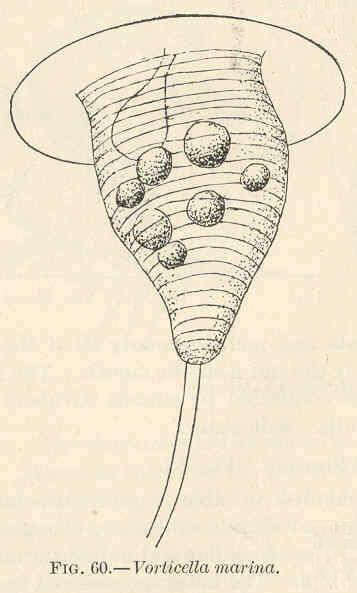
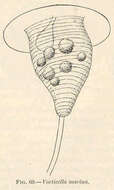
Vorticella marina.
-
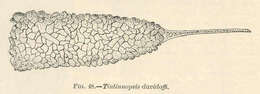
Tintinnopsis davidoffi.
-
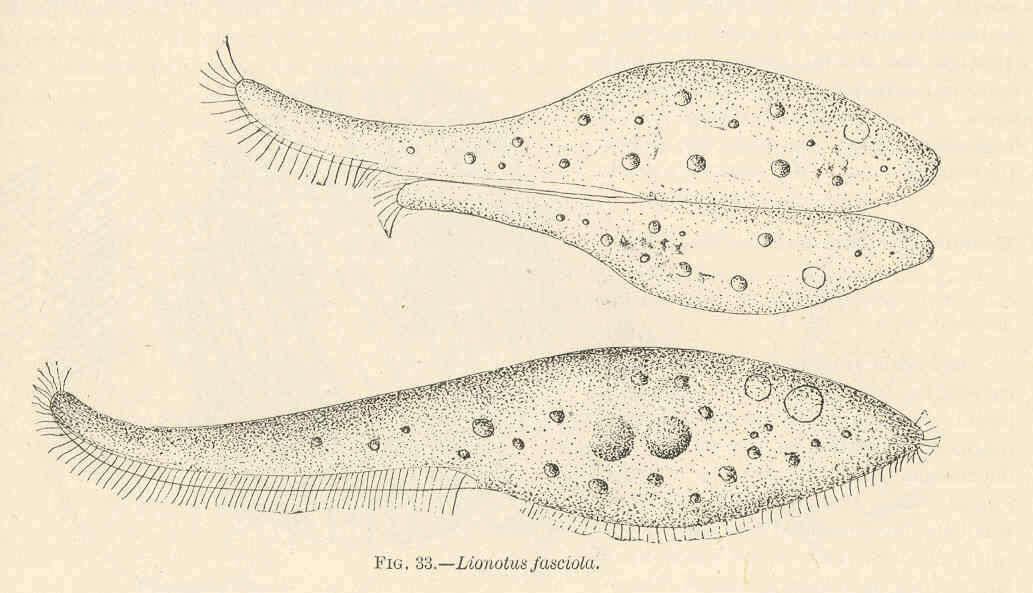
Lionotus fasciola.
-
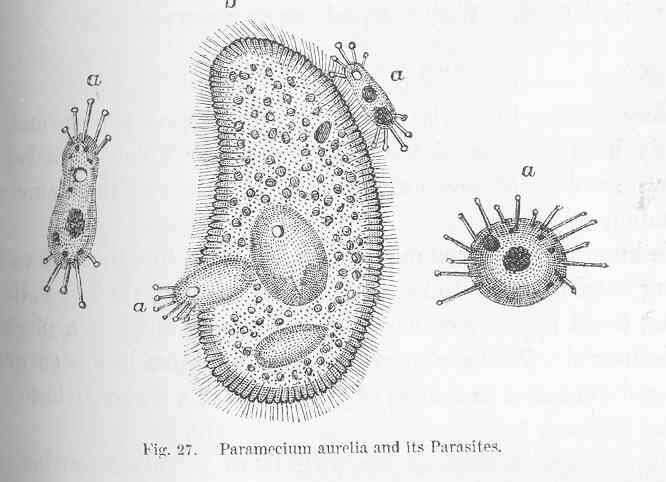
Paramecium aurelia and its Parasites.
-
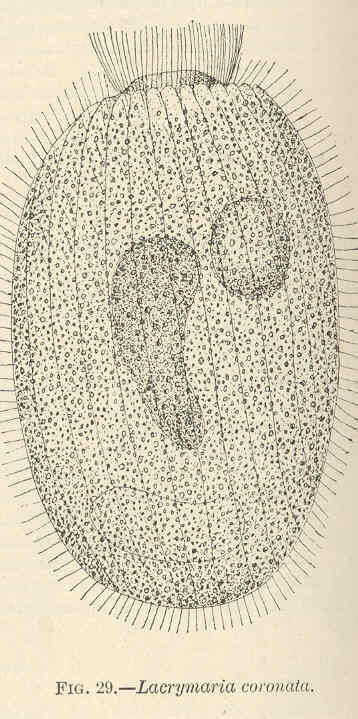
Lacrymaria coronata.
-
-
-